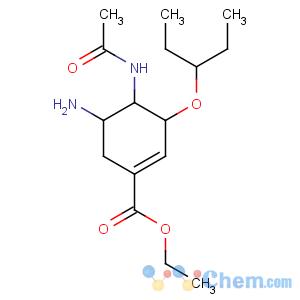
Title: Oseltamivir
CAS Registry Number: 196618-13-0
CAS Name: (3
R,4
R,5
S)-4-(Acetylamino)-5-amino-3-(1-ethylpropoxy)-1-cyclohexene-1-carboxylic acid ethyl ester
Molecular Formula: C16H28N2O4
Molecular Weight: 312.40
Percent Composition: C 61.51%, H 9.03%, N 8.97%, O 20.49%
Literature References: Orally active inhibitor of influenza virus neuraminidase; converted
in vivo to the active acid metabolite,
GS-4071. Prepn: N. W. Bischofberger
et al., US 5763483 (1998 to Gilead Sci.). Degradation kinetics: R. Oliyai
et al., Pharm. Res. 15, 1300 (1998). Bioavailability and pharmacokinetics: W. Li
et al., Antimicrob. Agents Chemother. 42, 647 (1998). Review of structure-activity studies and clinical development: C. U. Kim,
Med. Chem. Res. 8, 392-399 (1998). Clinical trial in prevention of influenza: F. G. Hayden
et al., N. Engl. J. Med. 341, 1336 (1999); in treatment of acute influenza: J. J. Treanor
et al., J. Am. Med. Assoc. 283, 1016 (2000). Review of potential use in influenza pandemic: P. Ward
et al.,
J. Antimicrob. Chemother. 55, Suppl. S1, i5-i21 (2005).
Properties: Pale solid. pKa 7.7 (25°); 6.6 (70°).
pKa: pKa 7.7 (25°); 6.6 (70°)
Derivative Type: Phosphate
CAS Registry Number: 204255-11-8
Manufacturers' Codes: GS-4104
Trademarks: Tamiflu (Roche)
Molecular Formula: C16H28N2O4.H3PO4
Molecular Weight: 410.40
Percent Composition: C 46.83%, H 7.61%, N 6.83%, O 31.19%, P 7.55%
Properties: White crystalline solid.
Therap-Cat: Antiviral (influenza).
Keywords: Neuraminidase Inhibitor; Antiviral; Sialic Acid Analogs.