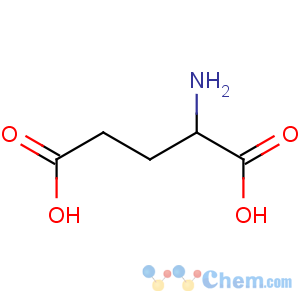
Title: Glutamic Acid
CAS Registry Number: 56-86-0
CAS Name: L-Glutamic acid
Synonyms: Glu; E; glutaminic acid; (
S)-2-aminopentanedioic acid; a-aminoglutaric acid; 1-aminopropane-1,3-dicarboxylic acid
Trademarks: Glutacid (Schering-Plough); Glutaminol (Syntex)
Molecular Formula: C5H9NO4
Molecular Weight: 147.13
Percent Composition: C 40.82%, H 6.17%, N 9.52%, O 43.50%
Literature References: Non-essential amino acid for human development; referred to as an excitatory amino acid (EAA) due to its role in neurotransmission. Isoln from wheat gluten: H. Ritthausen,
J. Prakt. Chem. 99, 454 (1866). Early chemistry and biochemistry:
Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) 950 pp.,
passim; J. P. Greenstein, M. Winitz,
Chemistry of the Amino Acids vols 1-3 (John Wiley and Sons, Inc., New York, 1961) pp. 1929-1954,
passim; C. W. Huffman, W. G. Skelly,
Chem. Rev. 63, 625-644 (1963). Interconversion from L-proline: S. Yoshifuji
et al., Tetrahedron Lett. 21, 2963 (1980). Determn in serum: C. D. Stalikas
et al., Eur. J. Clin. Chem. 32, 767 (1994); of naturally occurring levels in food: D. H. Daniels
et al., Food Addit. Contam. 12, 21 (1995). Identification as excitatory neurotransmitter: D. R. Curtis
et al., J. Physiol. 150, 656 (1960). Review of biosynthesis: A. Hamberger
et al., Adv. Biochem. Psychopharmacol. 27, 115-126 (1981); of metabolism and associated disorders: S. B. Prusiner,
Annu. Rev. Med. 32, 521-542 (1981). Review of receptor binding: P. A. Briley
et al., Mol. Cell. Biochem. 39, 347 (1981). Review as neurotransmitter: B. Engelsen,
Acta Neurol. Scand. 74, 337-355 (1986); E. Marmo,
Med. Res. Rev. 8, 441-458 (1988). Review of role in learning and memory: W. J. McEntee, T. H. Crook,
Psychopharmacology 111, 391-401 (1993); in Parkinson's disease: M. S. Starr,
Synapse 19, 264-293 (1995).
Books: R. Powell,
Monosodium Glutamate and Glutamic Acid (Noyes Dev. Corp., Park Ridge, N.J., 1968) 256 pp.;
Glutamic Acid: Advances in Biochemistry and Physiology L. J. Filer, Jr.
et al., Eds. (Raven, New York, 1979).
Properties: Orthorhombic, bisphenoidal crystals from aq alc. d420 vac 1.538. Melts 160° with conversion to L-pyrrolidonecarboxylic acid. Sublimes at 200°. [a]D22.4 +31.4° (6
N HCl). pK1 2.19; pK2 4.25; pK3 9.67. Soly in water (g/l); 8.64 (25°); 21.86 (50°); 55.32 (75°); 140.00 (100°). Insol in methanol, ethanol, ether, acetone, cold glacial acetic acid and common neutral solvents.
pKa: pK1 2.19; pK2 4.25; pK3 9.67
Optical Rotation: [a]D22.4 +31.4° (6
N HCl)
Density: d420 vac 1.538
Derivative Type: Hydrochloride
CAS Registry Number: 138-15-8
Trademarks: Acidulin (Lilly); Hypochylin (Recip)
Molecular Formula: C5H9NO4.HCl
Molecular Weight: 183.59
Percent Composition: C 32.71%, H 5.49%, N 7.63%, O 34.86%, Cl 19.31%
Properties: Orthorhombic bisphenoidal plates, dec 214°.
Derivative Type: Sodium salt
see Monosodium Glutamate
Derivative Type: Magnesium salt hydrobromide monohydrate
Synonyms: Magnesium glutamate hydrobromide; magnesium bromoglutamate
Trademarks: Psicosoma (Ferrer); Psychoverlan (Verla)
Molecular Formula: C10H17BrMgN2O8.H2O
Molecular Weight: 415.47
Percent Composition: C 28.91%, H 4.61%, Br 19.23%, Mg 5.85%, N 6.74%, O 34.66%
Derivative Type: DL-Form
CAS Registry Number: 617-65-2
Properties: Orthorhombic crystals from water, 225-227° (dec). dD20 1.4601. Soly in water (g/l): 20.54 (25°); 49.34 (50°); 118.6 (75°); 284.9 (100°). Sparingly sol in alcohol, ether, petr ether.
Density: dD20 1.4601
Derivative Type: D-Form
CAS Registry Number: 6893-26-1
Properties: Shiny leaflets from water. [a]D20 -30.5° (c = 1.00 in 6
N HCl).
Optical Rotation: [a]D20 -30.5° (c = 1.00 in 6
N HCl)
Therap-Cat: Nutritional supplement. Hydrochloride as gastric acidifier. Magnesium salt hydrobromide has been used as anxiolytic.
Keywords: Replenishers/Supplements; Nutrient.