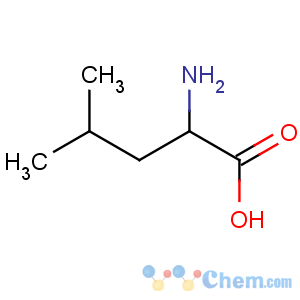
Title: Leucine
CAS Registry Number: 61-90-5
CAS Name: L-Leucine
Synonyms: Leu; L; 2-amino-4-methylvaleric acid; a-aminoisocaproic acid; (
S)-2-amino-4-methylpentanoic acid
Molecular Formula: C6H13NO2
Molecular Weight: 131.17
Percent Composition: C 54.94%, H 9.99%, N 10.68%, O 24.39%
Literature References: An essential amino acid for human development. Discovery of leucine is attributed to Proust in 1819 who reported its separation from fermented milk curds. Early chemistry and biochemistry:
Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) 950 pp.,
passim; J. P. Greenstein, M. Winitz,
Chemistry of the Amino Acids vols 1-3 (John Wiley and Sons, Inc., New York, 1961) pp. 2075-2096,
passim. Chromatographic determn of specific activity in biological fluids: E. P. Donahue
et al., J. Chromatogr. 571, 29 (1991); separation of isotopes: P. Q. Baumann
et al., ibid. 573, 11 (1992). Metabolism in humans: K. J. Motil
et al., Metabolism 30, 783 (1981); in children receiving parenteral nutrition: O. Goulet
et al., Am. J. Physiol. 265, E540 (1993). Kinetics modelling in humans: C. Cobelli
et al., ibid. 261, E539 (1991).
Properties: White glistening hexagonal plates from aq alc. d18 1.293. Sublimes at 145-148°. Dec 293-295° (rapid heating, sealed tube). [M]D +21.0° (5
N HCl); [M]D +29.5° (glacial acetic acid). [a]D25 -10.8° (c = 2.2); [a]D26 +15.1° in 6
N HCl (38 mols HCl per mol leucine); [a]D20 +7.6° in 3
N NaOH (30 mols NaOH per mol leucine). R
f value 0.79. Soly in water (g/l): 22.7 (0°); 24.26 (25°); 28.87 (50°); 38.23 (75°); 56.38 (100°); in 99% alcohol: 0.72; in acetic acid: 10.9. Insol in ether.
Optical Rotation: [a]D25 -10.8° (c = 2.2); [a]D26 +15.1° in 6
N HCl (38 mols HCl per mol leucine); [a]D20 +7.6° in 3
N NaOH (30 mols NaOH per mol leucine)
Density: d18 1.293
Derivative Type: DL-Form
Properties: Leaflets fom water. Sweet taste. Dec 332° (also reported as 290°). Sublimes. pK1 2.36; pK2 9.60. Soly in water (g/l): 7.97 (0°); 9.91 (25°); 14.06 (50°); 22.76 (75°); 42.06 (100°); in 90% alcohol: 1.3. Insol in ether.
pKa: pK1 2.36; pK2 9.60
Derivative Type: Hydrochloride
Molecular Formula: C6H13NO2.HCl
Molecular Weight: 167.63
Percent Composition: C 42.99%, H 8.42%, N 8.36%, O 19.09%, Cl 21.15%
Properties: Crystals. Freely sol in water.
Use: Nutrient.