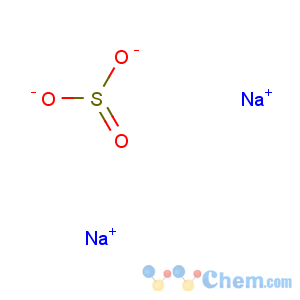
Title: Sodium Sulfite
CAS Registry Number: 7757-83-7
Molecular Formula: Na2O3S
Molecular Weight: 126.04
Percent Composition: Na 36.48%, O 38.08%, S 25.44%
Line Formula: Na2SO3
Properties: Small crystals or powder. It is fairly stable and does not oxidize as readily as the hydrated sulfite. Sol in 3.2 parts water; sol in glycerol; almost insol in alcohol. pH about 9.
Keep well closed. LD50 i.v. in mice: 175 mg/kg, Hoppe, Goble,
J. Pharmacol. Exp. Ther. 101, 101 (1951).
Toxicity data: LD50 i.v. in mice: 175 mg/kg, Hoppe, Goble,
J. Pharmacol. Exp. Ther. 101, 101 (1951)
Derivative Type: Heptahydrate
Properties: Efflorescent crystals. Unstable, oxidizing in the air to sulfate. Sol in 1.6 parts water, about 30 parts glycerol; sparingly sol in alc. The aq soln is alkaline and dissolves sulfur. pH ~9.
Keep well closed in a cool place. Note: The commercial heptahydrate (about 90% pure, the remainder consisting chiefly of the sulfate) has been largely replaced by the commercial anhydr salt (about 96-99% pure) which is more stable.
Use: Chiefly in photographic developers and instead of "hypo" for fixing prints; bleaching wool, straw, silk; generating SO2; as reducer in manuf dyes; silvering glass; removing traces of Cl in bleached textiles and paper; preserving meat, egg yolks, etc.